Welsh Government – ATMP Development and Manufacturing Facility
The Opportunity
Austin were appointed by The Welsh Government (WG) to undertake the detailed design of a new Advanced Therapeutic Medicinal Products research, development and manufacturing facility.
The design had to address constraints of the existing building including external wall profiles, internal structure locations, relatively low floor to floor heights and existing plant and HVAC provision whilst providing future flexibility and adaptability.
A key aspect of the design was to provide cGMP compliance for the lab areas and suitably efficient layout for material and waste flows.
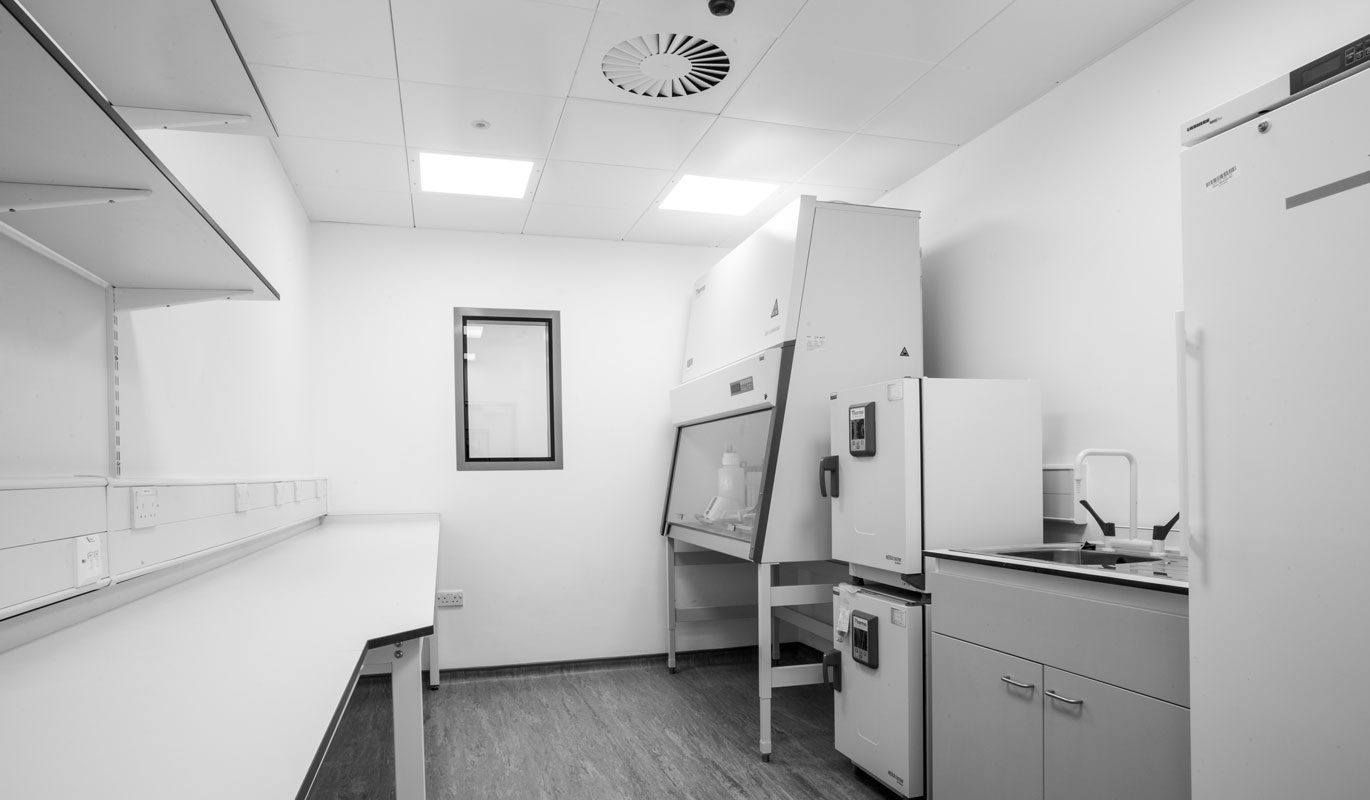
The brief extended to include the provision of modern and functional spaces to best respond to the specific needs of this type of facility.
The design solution responded to the Client’s need for a new 3,000m² facility which is designed to cater for future adaptability to suit differing needs and possible alternative trends.
Initially the new facility was to be operated by ReNeuron, a leading research company specialising in the development of novel stem cell therapies including clinical stage development.
The overall implementation phase was to be such that phase 1, R&D Laboratories, Process Development area and QC laboratories were to be delivered as one phase and the Modular Small Scale Manufacturing areas to be designed as Grade B, C and D as Phase 2.
The Solution
Austin commenced with challenging requirements where by the solution included offices to provide support functions with a modern environment. The fit out and design approach provides a ‘high-tech’ and professional image, in compliance with the vision and mission of ReNeuron and WG.
- New QC Laboratories for use as development laboratories initially and to support manufacturing in the future.
- The construction was based on using conventional plasterboard walls and clip in ceiling system, suitable for laboratories. Proprietary metal panelling systems for wall and ceilings were used in classified areas.
- The R&D Laboratories, Assay Development, Histology and Process Development, were designed for use for ReNeuron’s ongoing product and process research.
- A key aspect of the design for Phase 1 was based around suitable and complaint flows, material and waste flows, whilst providing bright, functional spaces to best respond to the specific needs of this type of facility.
- Phase 1 of the new facility was handed over earlier and was developed to provide a flexible work space which ReNeuron occupied to commence research and development work, facilitating the move from their existing facility.
“A massive thank you to all for your hard work and for getting us to this point. Everyone at ReNeuron is really excited about getting into their ‘new home’.” – Sharon Grimster, General Manager, ReNeuron.